Researchers hope nasal vaccine may block Covid transmission
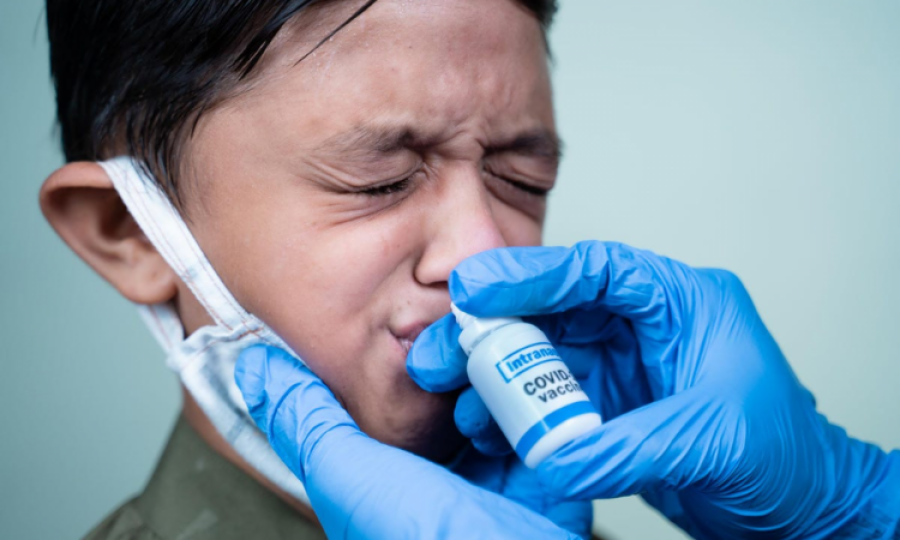
USA: As the primary purpose of the vaccines currently being used against Covid-19 is to save those infected with the virus from serious disease forms resulting in hospitalisation and death, a research team is trying to develop sterilising vaccine that can, through nasal administration, counter the virus at the entry point (nose), and interrupt the circle of contamination, blocking transmission between individuals.
Vaccination induces an immune response based on two types of cells: T lymphocytes, capable of destroying infected cells, and B lymphocytes, which produce antibodies capable of neutralising the virus (SARS-CoV-2 in the case of Covid-19) to prevent it from multiplying and infecting new healthy cells. Current vaccines are administered intramuscularly and thus called “systemic”in that they allow the activation of a pool of immune cells in the entire body, reaching infected organs through blood circulation.
Effective as it is, this systemic immunity does not allow the mobilisation of a high level of B and T lymphocytes in the nasal cavity and the lungs which would block the virus immediately upon arrival, promoting a rapid and effective protection.
Conversely, an intranasal vaccination induces not only a systemic but also a local immune response at the gate of the entry of SARS-CoV-2. The local activation of the immune cells of the nasal mucosa would thus make it possible to overcome the race against time between the virus multiplying in the respiratory system, and the systemic immune system. The mucosal vaccination would quickly stop the virus, limiting its dissemination and replication in the human body and thus preventing transmission and contamination.
It’s also worth noting that the immune cells it activates (resident T and B lymphocytes in the nose, mouth and upper respiratory tract) differ from those activated by the conventional intramuscular vaccination. Those B lymphocytes in turn produce particular antibodies, IgA (Type A immunoglobulins), which are only very weakly induced by the intramuscular route which mainly induces B cells producing IgG (Type G immunoglobulins).
IgAs have a greater capacity than IgGs to “capture” and neutralise viruses. Furthermore, they are more versatile than IgGs and thus can retain a significant level of effectiveness despite possible variations in the virus.For these reasons, mucosal vaccination would be better suited to block inter-individual transmission and achieve sterilising immunity.
The only example of intranasal vaccine available in human health is the FluMist vaccine. This flu vaccine is based on an attenuated form of the causative virus (influenza), approved in the United States and Europe, and has an efficacy that surpasses that of the intramuscular vaccine in young children.
Research team BioMAP laboratory, Joint University-INRAE ISP 1282 research unit, led by Prof Isabelle Dimier-Poisson, has recognised experience in immunology and mucosal vaccination. They have worked on an innovative anti-Covid mucosal vaccine strategy.
Their vaccine is based on three innovations. The antigen is an original fusion protein composed of the Spike (S) protein, associated with another protein of the virus, the nucleoprotein (N). This fusion strategy allows the vaccine to maintain its efficacy against different variants.
To optimise the activation of the mucosal immune response, the antigen is wrapped in “nano-carriers” that allow an optimal administration of the protein by sticking to the mucous membrane. In this manner there is no need for an adjuvant (likely to create inflammation), thus reducing the risk of side effects.
The last key element is a dedicated delivery system, a spray capable of depositing the vaccine in the nasal cavity, precisely at areas containing the mucosal immune cells.
Other teams are following the same approach to develop anti-Covid vaccine delivered through the mucosal route. CanSino Biologics’, a vaccine currently being tested in China, is administered by inhalation using a nebuliser. The second is iNCOVACC, developed by Bharat Biotech, for primary vaccination in two doses administered through the nose. Both pose a potential risk of inflammation. AstraZeneca also recently announced the disappointing results of its first clinical trials of a nasal vaccine.
A key point, which is underlined by the researchers of this latest vaccine candidate, is the importance of the spray system. So far the vaccine candidate has given excellent results against multiple variants of SARS-CoV-2 in gold-standard preclinical models (mice and hamsters) Clinical trials in humans are scheduled for the current year which would further validate its efficacy.
The challenge is to stay at the level of the nasal cavity in order to limit the risks of an uncontrolled immune reaction that can lead to an excessively strong inflammatory reaction in the lungs (“cytokine storms”). To ensure that, the vaccine needs to be completely deposited in the critical areas of the nose where the virus nests, and where the relevant immune cells are located.
A vaccine spray is different from a classical therapeutic spray designed for repeated use. It must deliver a single, very precise dose. The research team is collaborating with two companies specialised in intranasal delivery systems, Aptar pharma and Medspray, to develop the spray.
The potential effectiveness of the spray systems is assessed by two methods. In vitro, using an artificial model (nasal cast) which reproduces the human nasal cavity, and In vivo, by carrying out comparative
Trending
Popular
Dow University’s new rabies vaccine is just a phone call away
-
IRD role lauded in advancing ...
02:53 PM, 12 Mar, 2024 -
Over one billion people worldwide ...
09:48 AM, 5 Mar, 2024 -
'Artificial tongue' introduced to ...
03:38 PM, 4 Mar, 2024 -
Health Alert: Dried fruits found to ...
11:47 AM, 2 Mar, 2024




